The Onglyza lawsuit has become a significant legal matter for those affected by the medication’s potential risks. Approved by the FDA in 2009 for managing type 2 diabetes, Onglyza was marketed as an effective solution to help control blood sugar without causing weight gain. However, over time, serious concerns have emerged regarding its links to heart failure, pancreatitis, and even pancreatic cancer. As a result, many patients are now seeking compensation through legal action against AstraZeneca and Bristol-Myers Squibb, the drug’s manufacturers.
In this detailed guide, we will explore the side effects, legal claims, and compensation options related to Onglyza, providing essential information for those who may have been impacted. Whether you’re considering pursuing a lawsuit or simply want to understand the risks, this article will provide valuable insights into the ongoing legal battle surrounding Onglyza.
What Is Onglyza?
Onglyza (saxagliptin) is a prescription medication approved by the U.S. Food and Drug Administration (FDA) in 2009 to help adults with type 2 diabetes control blood sugar levels. It belongs to a class of drugs known as dipeptidyl peptidase4 (DPP4) inhibitors that improve glucose regulation by increasing hormone levels that stimulate insulin release.
Onglyza became widely used because, unlike some other diabetes drugs, it did not typically cause weight gain while effectively lowering blood sugar.
Why Lawsuits Were Filed Against Onglyza
While Onglyza helped manage blood glucose, research and safety reviews raised concerns that it could:
- Increase the risk of heart failure
- Be linked to pancreatitis
- Possibly increase pancreatic cancer risk
Heart Failure Risk
A major study published in The New England Journal of Medicine found that patients taking saxagliptin had a 27% higher rate of hospitalization for heart failure compared to patients taking a placebo. This was especially concerning for people with preexisting cardiovascular issues.
In April 2016, the FDA issued a Drug Safety Communication requiring stronger warnings on Onglyza’s label about the increased risk of heart failure.
Other Serious Side Effects
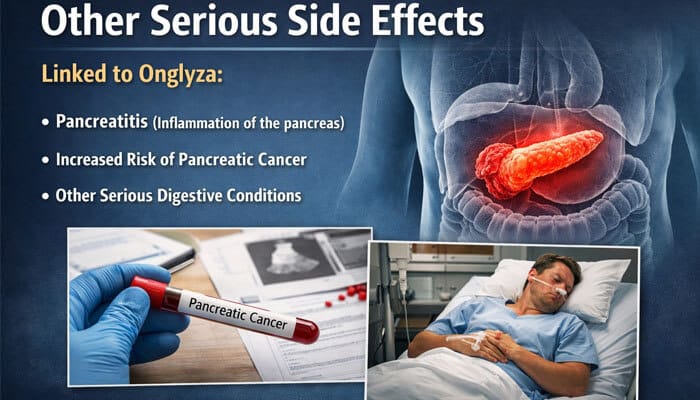
In addition to heart issues, lawsuits and warnings linked Onglyza to:
- Pancreatitis (inflammation of the pancreas)
- Increased risk of pancreatic cancer (in some studies)
- Other serious digestive conditions
The Legal Impact of FDA Warnings
The FDA Drug Safety Communication issued in 2016 was a significant turning point for Onglyza lawsuits. The FDA required stronger warning labels about the risks of heart failure linked to saxagliptin. This communication directly influenced ongoing litigation and became a cornerstone for plaintiffs’ claims, as they argued that AstraZeneca and BristolMyersSquibb failed to adequately inform patients and doctors about these risks.
Other Diabetes Medications and Lawsuits
Comparing Onglyza to other diabetes medication lawsuits helps understand the broader scope of pharmaceutical litigation.
- Januvia Lawsuit: Like Onglyza, Januvia (sitagliptin) has faced similar lawsuits due to cardiovascular risks and potential links to pancreatic issues.
- Farxiga Lawsuit: While not as widely litigated as Onglyza, Farxiga (dapagliflozin) also has some related claims, though it targets a different mechanism of action.
These lawsuits underscore the growing concerns within the DPP4 inhibitors class of drugs and their possible side effects. Understanding the similarities and differences in the legal outcomes of these drugs can shed light on the challenges facing plaintiffs in Onglyza lawsuits.
Class Action vs Individual Lawsuit: Which Option Is Best for Onglyza Victims?
When considering legal action, plaintiffs often wonder whether to pursue a class action lawsuit or an individual claim.
- Class Action Lawsuits: These are often more efficient and can result in quicker settlements, but they may not provide the level of compensation or attention that individual lawsuits can achieve.
- Individual Lawsuits: These offer plaintiffs more control over their case and potentially higher compensation, but they can take longer and require more legal effort.
Detailed Legal Process: From Consultation to Settlement
Understanding the legal process for an Onglyza lawsuit is crucial for potential plaintiffs. Here’s a step-by-step guide:
- PreLitigation Steps: Attorneys assess the merits of your case and determine if you meet the legal criteria.
- Filing the Lawsuit: Once the case is deemed viable, the attorney files a lawsuit in the appropriate jurisdiction.
- Court Process: The discovery phase, where both sides exchange evidence, is followed by trial preparations.
- Settlement Negotiation: Many cases settle before reaching trial, often resulting in compensation for the plaintiff.
Recent Legal Precedents: Key Onglyza Lawsuit Cases and Court Rulings
As of 2025–2026, key court rulings have shaped the direction of Onglyza lawsuits. Some cases have seen dismissals due to expert testimony exclusion, while others are still in progress.
- 2025 Case Example: A major ruling in Eastern District of Kentucky dismissed several claims on the grounds that expert testimony was unreliable.
- Ongoing Cases: Many lawsuits are still being filed, with plaintiffs’ attorneys hoping to leverage new medical evidence connecting Onglyza use with heart failure and cancer risks.
Expert Testimony and Medical Evidence in Onglyza Lawsuits

In pharmaceutical injury cases, expert testimony plays a critical role.
- Medical experts often provide testimony to establish the link between Onglyza and the heart failure or pancreatic conditions suffered by the plaintiff.
- Scientific Evidence: Many lawsuits rely on recent clinical studies that reinforce the drug’s potential risks.
Emotional and Psychological Impact of Injuries
The psychological and emotional toll caused by serious health conditions related to Onglyza is often overlooked. Many individuals report anxiety, depression, and distress due to their ongoing health struggles.
- Psychological Damages: Some lawsuits claim damages for the emotional toll resulting from living with heart failure or recovering from pancreatitis.
How to Find a Lawyer for Onglyza Lawsuit
Choosing the right attorney for an Onglyza lawsuit is crucial for achieving success.
- What to Look For: Experience with pharmaceutical cases, a proven track record, and contingency fee arrangements.
- Consultation: Many law firms offer free consultations, so you can get expert advice without upfront costs.
Conclusion
The Onglyza lawsuit remains a significant mass tort, with heart failure and pancreatitis risks at the center of litigation. While some cases have faced challenges, many affected individuals may still pursue legal claims. For those who believe they were harmed by the drug, it is crucial to act quickly to meet statutes of limitations and gather the necessary medical records for an evaluation.
Onglyza lawsuit FAQs
1. What is the Onglyza lawsuit?
The Onglyza lawsuit involves legal claims against AstraZeneca and Bristol-Myers Squibb, alleging that Onglyza caused serious health risks like heart failure, pancreatitis, and pancreatic cancer due to inadequate warnings about its dangers.
2. What health risks are linked to Onglyza?
Onglyza has been linked to significant health complications, including heart failure, pancreatitis (inflammation of the pancreas), pancreatic cancer, and kidney problems, leading many patients to seek compensation.
3. Who can file a lawsuit for Onglyza?
Individuals who took Onglyza and experienced heart failure, pancreatitis, pancreatic cancer, or other related health conditions may be eligible to file a lawsuit against the drug’s manufacturers.
4. How does the FDA impact the Onglyza lawsuit?
In 2016, the FDA issued a warning about heart failure risks linked to Onglyza. This official Drug Safety Communication led to an increase in lawsuits filed by patients who suffered adverse health effects.
5. How long do I have to file an Onglyza lawsuit?
The time limit to file a lawsuit, known as the statute of limitations, varies by state. Generally, you have a few years from the diagnosis or from the point you became aware of the drug’s risks to file a claim.
Disclaimer: This article is for informational purposes only and does not constitute legal advice. If you or a loved one has been affected by Onglyza, please consult a qualified attorney for personalized legal guidance.


